過去最高の室温熱電変換性能指数を示す酸化物を実現
~安定で実用的な熱電変換材料の実現に大きな期待~
ポイント
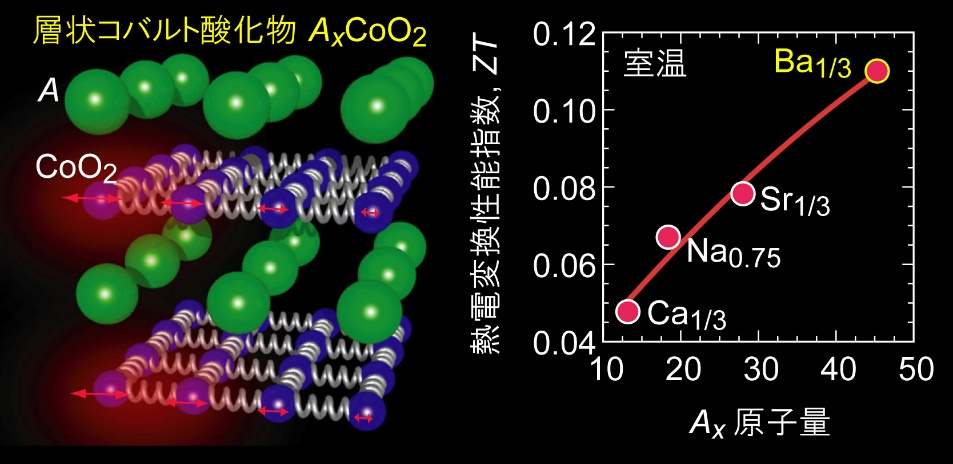
- 室温において過去最高の熱電変換性能指数ZT = 0.11を示す層状コバルト酸化物を実現。
- 層間イオンを重くすることで,電気特性を損なわずに低熱伝導率化することに成功。
- 安定で実用的な熱電変換材料の実現に大きな期待。
概要
北海道大学電子科学研究所の張 雨橋外国人客員研究員(JSPS外国人特別研究員)と太田裕道教授らの研究グループは,室温において過去最高の熱電変換性能指数ZT*1= 0.11を示す層状コバルト酸化物*2を実現しました。熱電変換は,工場や自動車から排出される廃熱を再資源化する技術として注目されています。熱電材料として,PbTeなどの金属カルコゲン化物*3が知られていますが,熱的・化学的安定性や,毒性があるという問題があり,大規模な実用化に至っていません。金属カルコゲン化物と比較して,多くの金属酸化物は高温・空気中においても安定していることから,日本では25年ほど前から熱電材料として研究されてきました。熱電材料の変換性能は,性能指数ZT [= (熱電能*4)2×(導電率*5)×(絶対温度*6)÷(熱伝導率)]で表され,金属カルコゲン化物の代表であるp型PbTeの室温におけるZTは約0.1です。金属酸化物の中でも,ナトリウムイオンを層間に含む層状コバルト酸化物は,熱電能が大きく,導電率も高いことから,実用的な熱電材料として期待されましたが,熱伝導率が高いため,室温のZTは0.03程度にとどまっていました。研究グループは,層状コバルト酸化物のナトリウムイオンを様々な金属イオンに置き換えて,金属イオン層が重くなるにつれて,電気的な特性は変化せず,熱伝導率だけが減少することを突き止め,最終的に重いバリウムイオンに置き換えたとき,室温でZTが0.11に達することを発見しました。一般に,性能指数ZTは高温になるほど上昇することから,安定で実用的な熱電変換材料の実現が期待されます。なお,本研究成果は,日本時間2020年10月13日にJournal of Materials Chemistry A誌に「Advanced Manuscript」としてオンライン掲載されました。
論文情報
論文名
Layered cobalt oxide epitaxial films exhibiting thermoelectric ZT = 0.11 at room temperature(室温で熱電変換性能指数ZT = 0.11を示す層状コバルト酸化物エピタキシャル薄膜)
著者名
高嶋佑伍(修士課程2年)1,張 雨橋2,魏 家科3,馮 斌3, 幾原雄一3, ジョヘジュン1,2, 太田裕道1,2(1北海道大学大学院情報科学院,2北海道大学電子科学研究所,3東京大学大学院工学系研究科)
雑誌名
Journal of Materials Chemistry A(英国・王立化学会 材料化学の専門誌, IF = 11.301)
DOI
公表日
日本時間2020年10月13日(火)に「Accepted Manuscript」としてオンライン出版されました。
背景
熱電変換は,工場や自動車から排出される廃熱を再資源化する技術として注目されています。熱電材料として,PbTeなどの金属カルコゲン化物が知られていますが,熱的・化学的安定性や,毒性があるという問題があり,大規模な実用化に至っていません。金属カルコゲン化物と比較して,多くの金属酸化物は高温・空気中においても安定であることから,日本では25年ほど前から熱電材料として期待され,研究されてきました。熱電材料の変換性能は,性能指数ZT [= (熱電能)2×(導電率)×(絶対温度)÷(熱伝導率)]で表され,金属カルコゲン化物の代表であるp型PbTeの室温におけるZTは約0.1です。金属酸化物の中でも,1997年に早稲田大学理工学部寺崎一郎助教授(当時。現名古屋大学理学研究科教授)らの研究グループによって見出された,ナトリウムイオンを層間に含む層状コバルト酸化物は,熱電能が大きく,導電率も高いことから,実用的な熱電材料として期待されましたが,熱伝導率が高いため,室温のZTは0.03程度にとどまっていました。
研究手法
研究グループは,層状コバルト酸化物AxCoO2の熱伝導率を下げるための,図1に示す戦略を考えました。AxCoO2の結晶構造は,Aイオン層とCoO2層が交互に積層した層状構造です。熱電変換性能は層に沿った方向のほうが層に垂直方向よりも高いことが知られています。AxCoO2の層に沿った方向の熱伝導は,主としてCoO2層の原子の振動が伝播することで起こります。Aイオンが軽い場合,CoO2層の振動は減衰することなく伝播するので,熱伝導率は高くなりますが,Aイオンを重くするとCoO2層の振動はすぐに減衰するという仮説を立てました。
熱伝導を簡単に説明します。1個1個の原子の振動を「ばね」とすると,熱の伝播は「ばね」の伸縮の伝播と似ています。「ばね」がたくさん入っているマットレスがあったとしましょう。マットレスの上に赤ちゃんを乗せてもマットレスの「ばね」は影響を受けないのに対し,例えば大きな力士が乗れば「ばね」は縮んで動かなくなります。このような考察から,Aイオンを重くすることで低熱伝導率化できると予想しました。
研究グループは,まずNa0.75CoO2薄膜を作製し,次いでイオン交換法によってNa0.75を重さが異なるCa1/3,Sr1/3,Ba1/3に置換したAxCoO2薄膜を作製しました。作製した薄膜の構造解析の結果,最初に作製したNa0.75CoO2薄膜のNaイオンが,Ca1/3,Sr1/3,Ba1/3に置換できたことが分かりました。その後,室温における導電率,熱電能,および熱伝導率を計測しました。
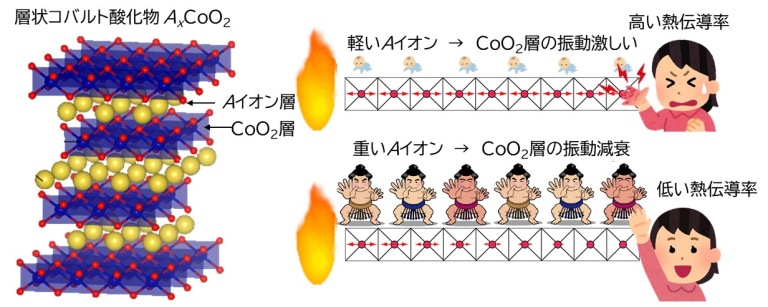
図1.層状コバルト酸化物AxCoO2の熱伝導率を下げるための戦略。(左)AxCoO2の結晶構造は,Aイオン層とCoO2層が交互に積層した層状構造です。(右)AxCoO2の層に沿った方向の熱伝導は,主としてCoO2層の原子の振動が伝播することで起こります。Aイオンが軽い場合,CoO2層の振動は減衰することなく伝播するので,熱伝導率は高くなりますが,Aイオンを重くするとCoO2層の振動はすぐに減衰します。1個1個の原子の振動を「ばね」とすると,熱の伝播は「ばね」の伸縮の伝播と似ています。「ばね」がたくさん入っているマットレスがあったとしましょう。マットレスの上に赤ちゃんを乗せてもマットレスの「ばね」は影響を受けないのに対し,例えば大きな力士が乗れば「ばね」は縮んで動かなくなります。このような考察から,Aイオンを重くすることで低熱伝導率化できると予想しました。
研究成果
図2に,室温において計測したAx置換AxCoO2薄膜の層に平行な方向の熱電特性をまとめて示します。導電率と熱電能はAイオンの置換によってわずかに影響を受けているように見えますが,[(熱電能)2×(導電率)]で表される出力因子はAxに依らずほぼ一定です。電流が流れるのはCoO2層なので,Axイオンの置換は電気的な特性には影響しないと言えます。これに対して,熱伝導率はAx原子量の増加に伴って単調に減少する傾向を示しました。Baはアルカリ金属とアルカリ土類金属の中から選ぶことができる最も重い元素です。この低熱伝導率化だけが熱電変換性能指数の変化にそのままに反映され,Ba置換したBa1/3CoO2では酸化物の室温の熱電変換性能指数としては最大の0.11に達することが分かりました。
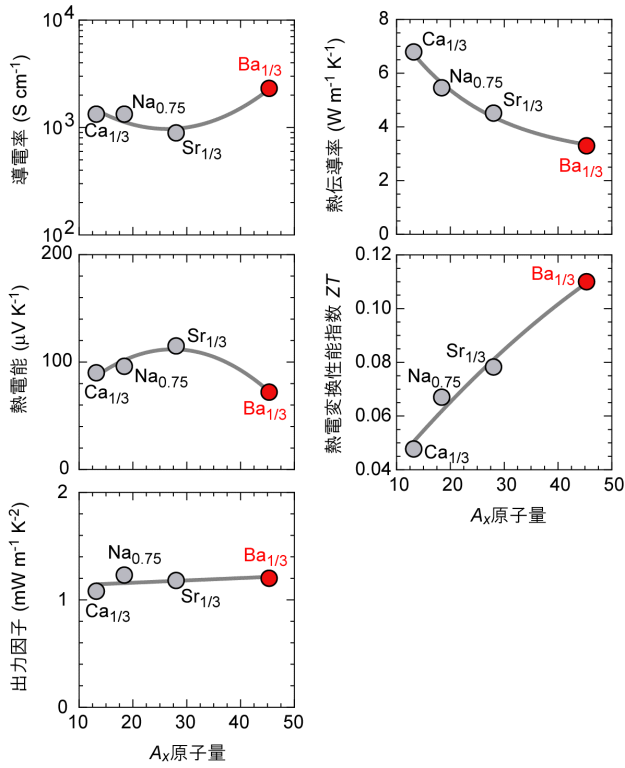
図2.Axを置換したAxCoO2薄膜の層に平行な方向の熱電特性(室温)。導電率と熱電能はAイオンの置換によってわずかに影響を受けているように見えますが,[(熱電能)2×(導電率)]で表される出力因子はAxに依らずほぼ一定です。電流が流れるのはCoO2層なので,Axイオンの置換は電気的な特性には影響しないと言えます。これに対して,熱伝導率はAx原子量の増加に伴って単調に減少する傾向を示しました。Baはアルカリ金属とアルカリ土類金属の中から選ぶことができる最も重い元素です。この低熱伝導率化だけが熱電変換性能指数の変化にそのままに反映され,Ba置換したBa1/3CoO2では酸化物の室温の熱電変換性能指数としては最大の0.11に達することが分かりました。
今後への期待
一般に,性能指数ZTは高温になるほど上昇します。現在,高温での熱電特性の計測も行っており,ZTが上昇することは確認済みです。今後,さらに組成を最適化するなど,熱電変換性能を増強することで,安定で実用的な熱電変換材料が実現すると期待されます。
謝辞
本研究は,日本学術振興会科学研究費助成事業・新学術領域研究(研究領域提案型)「機能コアの材料科学」(領域代表:松永克志 名古屋大学・教授)における計画研究「界面制御による高機能薄膜材料創製(課題番号 19H05791)」および「界面機能コア解析(課題番号 19H05788)」, 基盤研究(A)「熱電材料の高ZT化に向けたナノ周期平行平板構造の熱伝導率解明(課題番号17H01314)」,日本板硝子財団,イノベーション創出ダイナミック・アライアンス,物質・デバイス領域共同研究拠点の助成を受けた成果です。また,本研究の一部は,文部科学省委託事業ナノテクノロジープラットフォーム・東京大学微細構造解析プラットフォーム(課題番号JPMXP09A20UT0090)の支援を受けて行われました。
お問い合わせ先
北海道大学電子科学研究所 教授 太田裕道(おおたひろみち)
TEL 011-706-9428
FAX 011-706-9428
メール hiromichi.ohta[at]es.hokudai.ac.jp
URL https://functfilm.es.hokudai.ac.jp/
用語解説
*1 熱電変換性能指数ZT
熱電材料の熱→電気変換効率を示す指標。性能指数ZT [= (熱電能)2×(導電率)×(絶対温度)÷(熱伝導率)]で表される。室温付近で使用されるBi2Te3系の金属カルコゲン化物のZTは室温付近で約0.8,400℃程度の温度で使用されるPbTe系の金属カルコゲン化物のZTは室温付近で約0.1,400℃付近で約1。
*2 層状コバルト酸化物
酸化コバルトCoO2層とアルカリ金属またはアルカリ土類金属の層が交互に積層した結晶構造を有する酸化物結晶のこと。層状コバルト酸化物のひとつであるLiCoO2はリチウムイオン電池の正極活物質として広く実用化されている。
*3 金属カルコゲン化物
金属元素とカルコゲン元素(S,Se,Te)との化合物のこと
*4 熱電能
熱電材料に温度差を与えたときに発生する熱起電力(電圧)の温度係数のこと。
*5 導電率
電気の流れやすさを示す指標。
*6 熱伝導率
熱の伝わりやすさを示す指標のこと。












